Because Every Tooth Deserves Care.
Regenerative Dentistry: South Korea's Tooth Patch and the Road to Human Trials
Language :
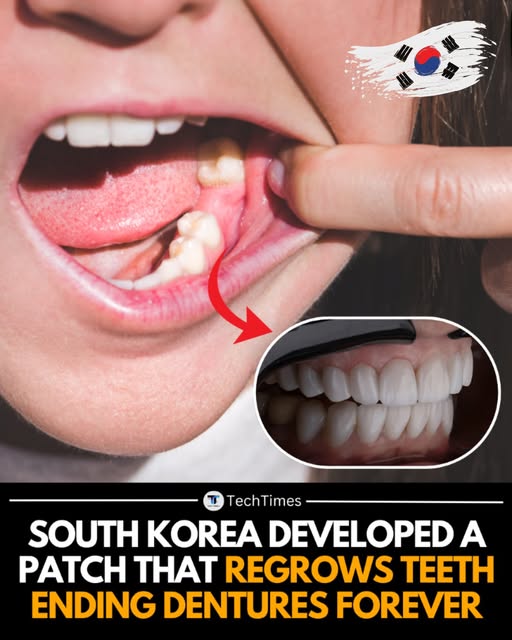
The Future is Now: South Korea's Tooth-Regenerating Patch
The prospect of naturally regrowing a damaged tooth is moving rapidly from science fiction to clinical reality, thanks to groundbreaking research from South Korea. Scientists there are developing a revolutionary tooth-regenerating patch designed to repair tooth defects, potentially replacing traditional fillings and expensive restorative procedures. This innovative technology aims to harness the body's own biological mechanisms to stimulate the regeneration of dentin and enamel.
What is the Regenerating Patch?
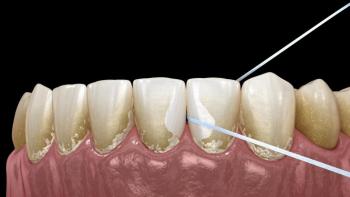
The "tooth patch" is a tiny, biocompatible material—often a scaffold embedded with special cells or growth factors—that is placed directly onto the damaged area of the tooth. Its primary function is to:
-
Serve as a Scaffold: Provide a structural framework for new dental pulp cells and surrounding tissues to migrate into and grow.
-
Deliver Signals: Release specific growth factors that encourage the surrounding dental stem cells to differentiate into odontoblasts (cells that form dentin) and potentially ameloblasts (cells that form enamel).
-
Seal the Defect: Act as a temporary sealant against oral bacteria, similar to a filling, while the natural healing process takes over.
If successful, this technology would offer a true biological solution, leading to a tooth that is structurally sound and integrated with the body, rather than being patched with synthetic materials like amalgam or composite resin.
The Animal Models: Testing Before Humans
Before any new medical or dental technology can be implemented in human patients, rigorous testing must be performed on animal models to establish safety, efficacy, and dosage. For the tooth-regenerating patch, researchers are primarily focused on animals whose dental structures or size are suitable for mimicking human procedures.
The two main animal models used in this type of regenerative dental research are:
-
Pigs (Swine Model):
-
Reason for Testing: Pig teeth, particularly their molar structure and dentition patterns, share significant anatomical and physiological similarities with human teeth. Their larger size allows surgeons to perform procedures and apply patches using techniques that closely resemble those used in a human clinical setting.
-
Role in Research: Pigs are often used in later-stage testing to confirm surgical feasibility and the quality of the regenerated tissue over longer periods.
-
-
Rats or Mice (Rodent Model):
-
Reason for Testing: While their tooth anatomy is less similar to ours, rodents are crucial for initial safety screenings and understanding the fundamental biological mechanisms of the patch. They have fast metabolisms and short lifecycles, making it easier to rapidly track the patch's cellular effects, toxicity, and gene expression changes.
-
Role in Research: Used for early-stage proof-of-concept studies and confirming that the embedded growth factors are not causing adverse systemic effects.
-
The Timeline: Aiming for 2030
The general goal for the South Korean researchers and similar teams worldwide is to move from large animal studies to human clinical trials.
-
Current Phase: Large animal studies (like the pig model) and optimization of the patch's material properties.
-
Next Phase (2025-2027): Submitting data for approval to begin Phase I and Phase II human clinical trials. These trials first test for safety and then check for efficacy on a small group of patients with dental defects.
-
Target Implementation (Post-2030): Full regulatory approval and widespread clinical implementation are often projected for the early 2030s, assuming the trials demonstrate clear superiority and safety over current treatments.
This innovation promises to revolutionize dentistry, potentially offering a future where a damaged tooth isn't drilled and filled, but simply guided to heal itself.
Critical Thinking: The Roadblocks to Perfecting the Tooth-Regenerating Patch
The South Korean research on the tooth-regenerating patch is revolutionary, but transitioning from successful animal trials (like those on pigs) to routine clinical practice by 2030 requires overcoming significant biological, technical, and regulatory hurdles. Applying critical thinking reveals several key areas where perfection is far from guaranteed:
1. Biological Control and Complexity
The primary challenge is reproducing the complex structure of a natural tooth.
-
Enamel vs. Dentin Regeneration:
-
Dentin regeneration (the layer beneath enamel) is relatively achievable because it involves healing within a confined space and activating nearby odontoblasts.
-
Enamel regeneration is vastly more complex. Enamel is formed by cells called ameloblasts, which are lost once the tooth fully erupts. The patch must not only stimulate the regeneration of this hard, acellular, and highly mineralized tissue but also somehow recreate the sophisticated enamel prisms—the microscopic structure that gives enamel its strength. This remains a major biological hurdle.
-
-
Pulp Integration and Vascularization: For the new dentin to be living, it must integrate perfectly with the existing dental pulp and allow for the growth of new blood vessels (vascularization) and nerve fibers. Failure here would result in a structurally sound but dead tooth (like a root-filled tooth), defeating the goal of true biological repair.
-
Immunological Reaction: Introducing a synthetic scaffold and foreign growth factors (like bone morphogenetic protein, BMP, or various fibroblast growth factors) always carries the risk of triggering an adverse immune response or chronic inflammation in the patient.
2. Technical and Manufacturing Scalability
Even if the patch works in a lab or on a pig, making it a viable product for millions of patients presents major technical issues.
-
Standardization: Teeth injuries are unique (location, depth, shape, severity). Creating a single "patch" that can effectively treat a tiny cavity on an incisor, deep decay on a molar, and root fracture simultaneously requires an incredibly versatile and easily adaptable material.
-
Clinical Handling: Dentists must be able to place the patch accurately and easily in the wet, contaminated environment of the mouth. The patch needs to be stable, adhesive, and quick-setting to ensure proper placement and immediate sealing of the damaged area.
-
Cost and Mass Production: Regenerative medicine is inherently expensive. For the patch to replace common, low-cost treatments like amalgam or composite fillings, the manufacturing cost must be low enough to achieve widespread adoption, a significant economic challenge in a high-volume dental market.
3. Regulatory and Ethical Roadblocks
The projected 2030 timeline is ambitious due to the stringent requirements of regulatory bodies (like the FDA in the U.S. or similar agencies globally).
-
Long-Term Efficacy: Regulators require data proving the patch not only works immediately but that the regenerated tissue is durable and stable for decades. Does the new enamel wear down faster? Does the new dentin become brittle over time? Answering these questions requires years of follow-up studies.
-
Safety Profile: The patch is considered a combination product (device + drug/biologic). It must pass three phases of human clinical trials, proving it is safe, effective, and free from causing tumors or uncontrolled cellular proliferation. Each phase can take several years.
-
Ethical Considerations: If the patch relies on autologous stem cells (cells harvested from the patient), the collection process (e.g., from wisdom teeth or bone marrow) adds complexity and cost, raising ethical questions about accessing and manipulating a patient's own tissue.
In conclusion, while the patch is a monumental step forward, achieving perfection by 2030 means successfully navigating the unforgiving complexity of biological regeneration, ensuring practical scalability for general practice, and satisfying the rigorous demands for long-term safety required by regulatory bodies.